Grid-scale storage technologies: technical view
This is the second post in a series on long-duration energy storage.
As mentioned in Part 1 of this post on grid-scale energy storage, pumped-storage hydropower (PSH) and lithium-ion (Li-ion) batteries are already deployed as storage technologies on the grid. So if we just install more of these, then that will be problem solved, right? The short answer is, unfortunately, no. Li-ion batteries are only really suited to short storage durations before they become too expensive to be favourable. And whilst PSH can be more economical for longer-durations, it is site-limited. In this technology deep-dive, we give a short introduction to some of the key existing and emerging technologies, and who the key players are in this space.
There are several key parameters that we need to consider in order to evaluate storage technologies:
- Rated power (W)
- Energy capacity (Wh)
- Round-trip efficiency (%)
- Levelised cost of storage per kWh (this will vary as a function of the above metrics)
Energy storage systems are typically defined by the rated power (or installed capacity) and the energy capacity. The rated power is measured in Watts (W) and measures the amount of power that can be generated instantaneously by a given source. The energy capacity is measured in Watt-hours (Wh). So a battery with a power rating of 10 MW and an energy capacity of 20 MWh can discharge all of its stored energy at maximum power in 2 hours (the discharge duration). The other key metrics we will consider here is the round-trip efficiency (or RTE), i.e. the proportion of energy (as a %) returned to the grid during a charge–discharge cycle. Discharge efficiency and cost per kWh were identified as the most important metrics for economic viability in this MIT study.
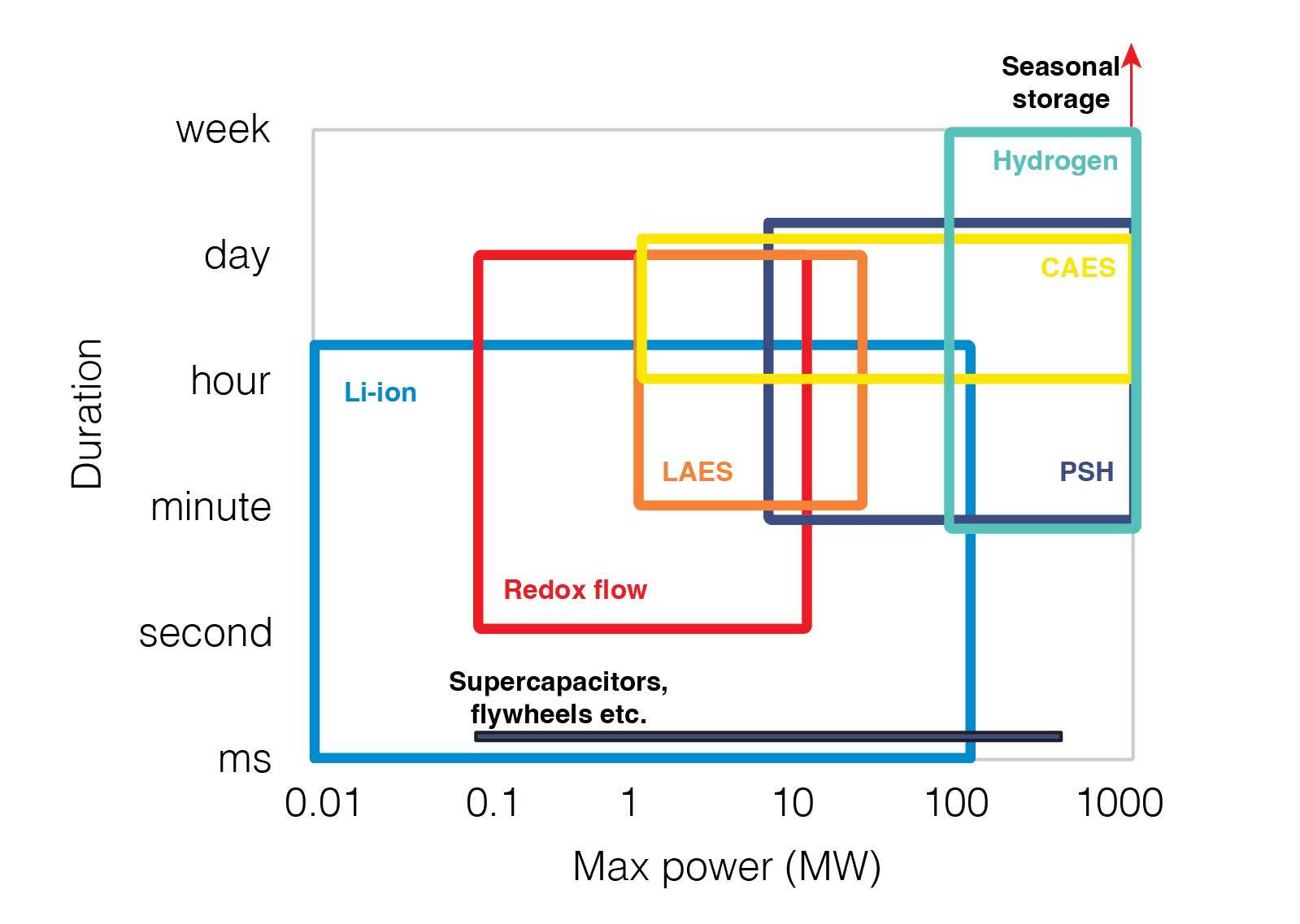
Typical power vs discharge duration for some of the technologies outlined here.
Established technologies
ELECTROCHEMICAL: Lithium-ion batteries
Li-ion batteries are already starting to be widely deployed as storage assets on the grid, with lithium iron phosphate (LFP) as the preferred cathode material. LFP is less energy dense than the lithium, nickel, manganese and cobalt (NMC) cathodes, which are preferred for electric vehicle applications, but benefits from the cheap and abundant raw materials of iron and phosphate.
The key advantages of Li-ion batteries are their short response times (on the order of milliseconds); their modularity and geographic flexibility—they can be added to homes, factories or act as grid scale storage units; they are also relatively cheap and have short build times. These attributes mean that to date Li-ion batteries have been used for ancillary services (frequency response) and load levelling, with typical installations with a duration (i.e. energy:power ratio) of one hour, with demand slowly increasing for two-hour and four-hour durations.
The main disadvantage for Li-ion batteries lies in the direct link between the power and energy capacities; since the energy capacity cannot be stored (and therefore scaled) separately, the cost of adding additional energy capacity becomes more or less linear.
The ability to use second-life EV batteries may bring costs down further—although this is unproven as a business model to date.
MECHANICAL: Pumped storage hydropower (PSH)
Pumped storage hydropower (PSH) has been used for over a century to provide additional energy in times of shortfall. Excess electricity is used to pump water from a lower to a high reservoir; when that electricity is required, the water is allowed to flow back to the lower reservoir through a turbine, thereby generating electricity. It is cheap (particularly for durations greater than 4 hours) but has fundamental limitations: it is geographically limited, most of the suitable sites have already been developed, and it has long regulatory and construction timeframes.
Companies: Quidnet Energy, RheEnergise.
MECHANICAL: Compressed-air energy storage
Compressed-air energy storage (CAES) is an established technology and although installations exist, it is not widely deployed due to lower round-trip efficiencies of conventional diabatic CAES (~50%) compared to PSH (~80%). The technology is similar to pumped hydro, but instead of pumping water uphill, air is compressed and stored in an underground cavern or storage unit. In order to generate electricity, the compressed air is then heated and expanded to turn a turbine. The advantages of CAES are its relatively lost cost, and the absence of toxic or pollutant side products since it only uses air.
Current installations (in Germany and Alabama) are diabatic CAES installations, which means that an external heat source (natural gas) is used to heat the air on evaporation. Adiabatic CAES recovers and stores the heat of compression–removing the need for natural gas and improving efficiencies to ~70%; whilst incurring the additional cost of including thermal energy storage.
Companies: Terrastor, Green-Y, Hydrostor, Hypnetic.
Emerging technologies
ELECTROCHEMICAL: Sodium-ion batteries
Sodium-ion batteries benefit from similar form and therefore production requirements as Li-ion batteries, they are a ‘drop-in’ replacement for Li-ion for grid storage. Their lower gravimetric energy density is not as much of a constraint in grid storage as it would be for use in EVs, so in the case of constraints to lithium supply (given the demand that will come from EV production), sodium-ion batteries are perhaps the most obvious alternative for grid storage.
The costs of sodium-ion batteries have the potential to be lower than Li-ion batteries due to (i) the use of sodium–it is very abundant and cheap; (ii) the use of aluminium rather than copper as the anode current collector (sodium does not alloy with copper). However, lower energy density requires bigger batteries, which can increase production costs.
Sodium-ion battery producers include Faradion, LiNa, Natron Energy, Altris, Tiamat and HiNa.
ELECTROCHEMICAL: Redox flow batteries (RFBs)
A redox-flow battery consists of a power stack and two circulating electrolyte solutions that are separated by an ion-exchange membrane as they pass through the stack. The electrolytes are pumped from the storage tanks through electrodes where the chemical energy is converted to electrical energy. Whilst they have less thermal runaway risk compared to Li-ion technologies, and so the cost of thermal management is lower, more cost is associated with the pumps and piping required to move the electrolyte.
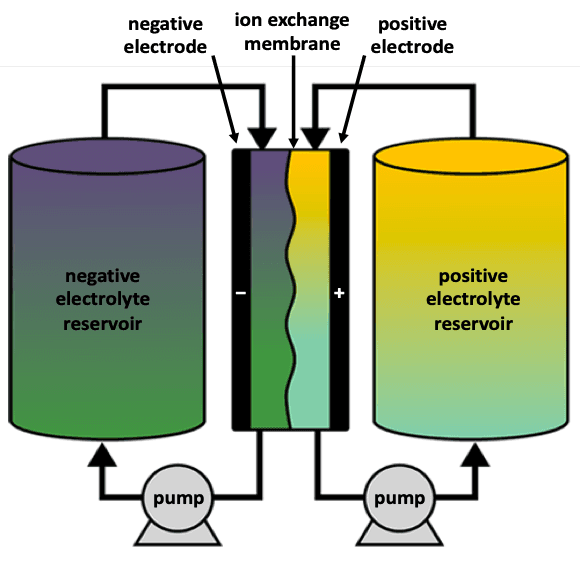
How a redox-flow battery operates: the electrolytes are held in storage reservoirs and are pumped through the stack where the chemical energy is converted to electrical energy. The energy:power ratio (discharge duration) can be increased for relatively low additional cost by increasing the size of the reservoirs. Source: BatteryBits
The most established type of redox flow battery is a vanadium flow battery, which exploits the varying oxidation states (charges) of vanadium—the electrolyte shuttles between V2+/V3+ or V4+/V5+ on charge/discharge. There are essentially infinite combinations of redox-flow-type systems depending on the chemical components. Organic redox flow systems (non-metal) are an emerging technology that could provide lower costs, but are earlier in the technology cycle and the electrolytes are more prone to degradation.
An important advantage of redox-flow batteries is that for a given power density the duration can be extended at relatively low cost by simply installing larger storage tanks to hold more electrolyte to increase the energy capacity; the power capacity is controlled by the size of the electrodes. Despite these fundamental attributes that make flow batteries technologically attractive for grid-scale storage, they remain too expensive. They are over 1.5 times as expensive as Li-ion technologies for short durations, and more expensive than CAES and PSH for longer discharge durations.
Companies in this space include ESS Tech Inc., RFC Power, Sinergy Flow, Volt Storage, Flux12, Quino Energy, Noon Energy, Agora Energy.
ELECTROCHEMICAL: Metal-air batteries
Metal-air batteries exploit the reversible energy changes associated with reacting a metal with oxygen in the air and cycling between the metal and its oxidised state. Form Energy are commercialising iron-air batteries by exploiting reversible ‘rusting’; the battery is charged by using energy to convert the iron oxide (rust) back to metallic iron, and on discharge the iron is then converted back to iron oxide. The use of cheap, and widely available iron, as the main chemical component is critical to lowering the cost of this technology to make it competitive with typical base load generation for multiday storage, Form Energy claim to be able to provide power for up to 100 hours. Iron-air technology is similar to well-established zinc-air battery chemistry, used in button cells and hearing aids; although to-date zinc-air batteries have not been rechargeable.
CHEMICAL: Hydrogen
At first glance, using hydrogen as a fuel to power the grid is extremely attractive; using renewable electricity and water to produce green hydrogen via electrolysis, which is then pumped into a fuel cell to generate electricity for the grid or burned to turn turbines in a generator.
There are two critical advantages for hydrogen. The first is that it can be stored in essentially unlimited quantities, since the ‘charge’ and ‘discharge’ processes are decoupled, (it could equally be thought of as a dispatchable fuel source). Secondly, it can be burned in a turbine using similar technology to that used in gas-fuelled power plants. However, hydrogen as storage has a round-trip efficiency of around 30%, due to compounding of 50-60% efficiency of typical electrolysers and fuel cells; this number is even lower if the hydrogen is burned in a turbine. This means that only a third of the energy used to generate the hydrogen will be returned to the grid when it is used.
Although projected to become much cheaper, hydrogen produced via electrolysis (green hydrogen) today costs between $4-7/kg of hydrogen, much higher than the $1-2/kg for grey hydrogen produced from methane. The proton electrolyte membrane (PEM) electrolysers that are best-suited to intermittent operation do not yet produce green hydrogen at scale and require expensive metals including platinum and iridium, which themselves could be subject to supply chain constraints and high prices. Add to this the fact that hydrogen is not currently transported or distributed at scale (it is typically used at the point of production in e.g. ammonia synthesis) and the challenge of scaling hydrogen for energy generation becomes clear. Nonetheless hydrogen- and ammonia-based power generation form an integral part of several energy security strategies going forward, notably that of Japan.
THERMAL: Liquid-air energy storage
Energy is stored in compressed and cooled liquid air; as the air is evaporated it turns turbines which generate electricity. The efficiency is improved if the excess heat generated on cooling is stored and used to heat the air as it is released.
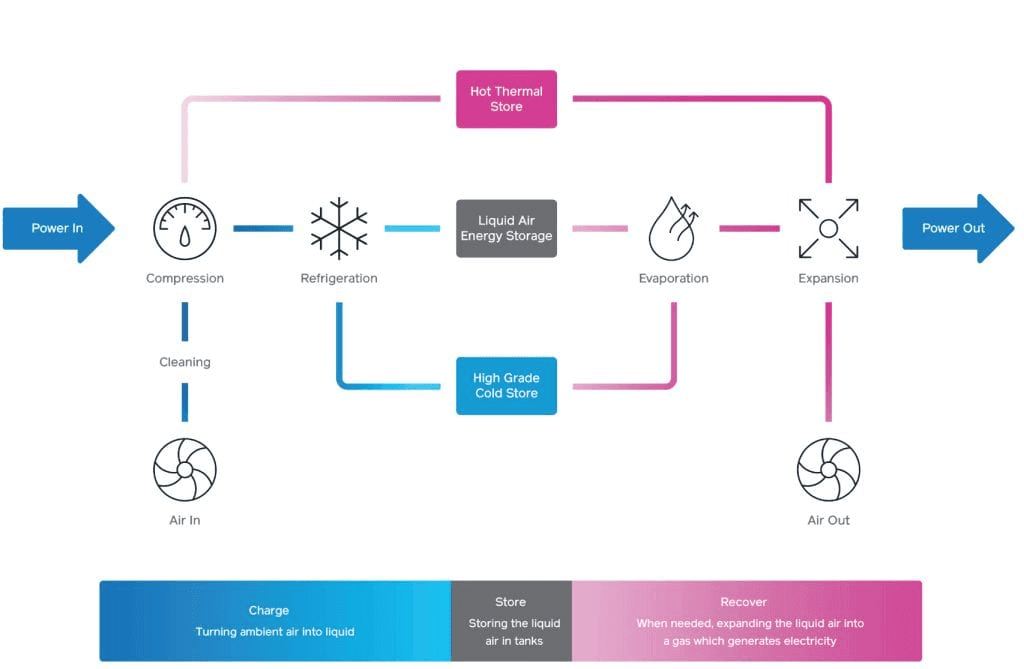
A schematic of how a liquid-air energy storage system operates: air is cooled and compressed, then stored at low temperature until electricity is required. The air is then heated and expanded to turn turbines and generate electricity. Storing of the heat generated on cooling results in a high round-trip efficiencies. Source: Highview Power
LAES is still in the early stages of commercial deployment and so is relatively expensive: it is the liquefaction technology that accounts for 45-70% of the cost (400-500 $/kWh for a 100MW / 400MWh installation) whereas the tanks required account for <10%. Changes to the heat carrier medium can reduce costs, at the expense of efficiency. An alternative is hybrid LAES (external heat sources for evaporation, either from fuel or from waste heat) that reduces costs to 300-400 $/kWh, at the expense of efficiency.
Carbon-dioxide-based compressed-air energy storage is an interesting development—since the boiling point of CO2 is much higher than that of nitrogen (the component of air that is liquified), it can be liquefied using moderate pressure.
Companies: Highview Power, phelas, Energy Dome (compressed-CO2).
THERMAL: Sensible/latent heat
Sensible heat technologies are still emerging and exploit the heat capacity of different materials to store heat. Storage media can consist of sand, concrete, molten salt and many other options. Although sand is cheaper than its molten salt equivalent (~$35/ton vs $950/ton), it has a worse specific heat capacity (830 J/kg/K vs 1545 J/kg/K) and therefore worse energy density.
Molten salt technologies—a type of sensible heat technology—are usually coupled with solar installations, such as concentrated-solar power (CSP), where heat from the sun is used to increase the temperature of molten salt. This heat is used to heat water to steam to turn turbines and generate electricity but it is also possible to use the thermal energy directly for heating. Either the heat is generated using a parabolic trough where the solar energy is concentrated to heat the salt directly, or alternatively solar-derived electricity is used to power a heat pump which then heats the salt.
By contrast, latent heat storage technologies exploit the thermodynamics of phase changes in materials: instead of relying on the heat capacity of the solid it makes use of the energy changes on going from solid to liquid and vice versa.
Companies: Malta, Hyme, Airthium, Polar Night Energy, RedoxBlox, Kraftblock, 1414 Degrees, Azelio
To summarise, the following table compares some of the technologies outlined above:
Technology | Max installation to date* | Approx. response time (s) | RTE (%) | Calendar life (yrs) | Advantages | Barriers to deployment |
|---|---|---|---|---|---|---|
Li-ion | 400 MW | 0.001 | 90 | 15 | Cheap | Li demand |
Redox flow (vanadium) | 100 MW 400 MWh Dalian, China | 0.1 | 65-70 | 20 | Can increase energy:power ratio | Cost |
CAES | 300 MW | 20 | 50 | 60 | Cheap (salt cavern storage) | Geographically limited |
PSH | 3.6 GW | 20 | 80 | 60 | Mature | Long build time |
Hydrogen | 150 GWh planned | 1 | 30 | 30 | Flexible | Low RTE |
LAES | 5 MW | 60 | 55 | 35 | Location flexible | Early tech |
Thermal (molten salt) | 510 MW 3 GWh Ouarzazate, Morocco | 60 | 50-60 | 30 | Scalable | Low energy density |
Low cost is key
In our previous post we identified three main energy-provision value streams for storage technologies: short duration discharge for peak-shaving, medium duration discharge for intraday load levelling and the multiday to seasonal large capacity and long duration discharge.
For short (and medium) durations, performance is key: the response time needs to be fast and the RTE relatively high to maximise the profits from arbitrage opportunities. However, just as peakers can be expensive because they are so responsive: the cost (levelised of storage or LCOS) per MWh can be relatively high. For Li-ion batteries it currently lies around $150/MWh. (The US Department of Energy’s energy storage cost and performance database is an excellent resource for exploring the costs and cost drivers of the different technologies, as a function of capacity and duration).
Lithium-ion batteries are extremely performant and well-suited to these shorter discharge duration opportunities. Given their ever-increasing penetration, new Li-ion-adjacent technologies remain an interesting area for investment e.g. second-life batteries for stationary storage, battery recycling, alternative mining. The extent to which alternative technologies can compete will depend on the extent of supply chain bottlenecks (either of critical minerals e.g. lithium/copper or of the battery units themselves) and their impact on battery prices, since Li-ion batteries may be prioritised for EVs and for higher-value frequency regulation services.
Low cost is the critical factor for longer duration discharge technologies. Here, the storage technologies are competing economically with base load generation–the marginal price of energy is now lower, and technologies must be aiming for a maximum LCOS of $50/MWh. Of existing technologies, CAES and PSH are cheap options that are currently available, but alternatives are needed because of the geographical constraints of both. Most of the suitable sites for PSH have already been developed and there are long lead times to build any new capacity. To keep costs low the technologies that perform best have (i) energy capacity and power capacity decoupled and (ii) low cost raw materials e.g. air for CAES or the iron in Form Energy’s iron-air batteries.
So what about hydrogen? Hydrogen has the lowest RTE (~30%) of all the technologies investigated, but the significant policy and financial support behind its deployment is accompanied by predictions of aggressive price decreases. If efficiency doesn’t impact cost significantly (say it is produced using very low cost curtailed renewables) then RTE isn’t necessarily important. However, there are risks associated with this assumption: (i) increased electrification will increase demand overall and (ii) smart grids and demand-side response efficiencies may raise base prices by smoothing demand.
In this blogpost we’ve given an overview of some of the technologies with the potential to provide storage across multiple timescales. Exactly which technologies will dominate (or what new technologies will be developed) remains to be seen and will be the focus of much investment activity over the coming years. But as we electrify large sectors of our economies to decarbonise them, the management and design of our electricity supply systems will be as critical as ever.
And if you're working on something exciting in this space or have comments on this piece – do get in touch!
References & further reading
Aurora Energy Research Long duration electricity storage in GB
Pacific Northwest National Laboratory Energy Storage Cost and Performance Database
BEIS Benefits of long-duration electricity storage
Long duration energy storage council