Ammonia: fertiliser and future fuel
11 min read
What is ammonia and what is it for?
Ammonia is essential to human life on earth. The size of the global population today would be much smaller without readily-available and cheap fertilisers that enabled the scale of food production required to feed the world—almost all of these nitrogen-based fertilisers are made from ammonia.
Chemically, ammonia is a humble four atom molecule composed of nitrogen and hydrogen with the formula NH₃. And it is as ammonia (or related derivatives) that plants get the nitrogen they need. Nitrogen is critical for plant growth—it is an essential component of amino acids and DNA—but the nitrogen gas N₂ in the air has to be converted to a more reactive form, such as ammonia, before it can be taken up by plants. This is a challenging and energy intensive process because nitrogen gas is so stable; the two nitrogen atoms are linked by a triple bond which requires a lot of energy to break. In nature this is performed by the enzyme nitrogenase present in microorganisms in the soil.
The industrial scale-up of the production of ammonia for fertilisers—the Haber-Bosch process—was designed in the early 20th century and it is estimated that 50% of all nitrogen in the human body has been through this process(!).
Why is it a climate problem?
Ammonia production is an energy and emissions intensive process. It requires 8.6 EJ of energy and accounts for 2% of global energy consumption. It is responsible for 450 MtCO₂e of direct emissions (90% of which are from the hydrogen production) as well as indirect emissions from electricity generation for nitrogen separation from air.
To understand where these emissions come from, we need to understand how it is made in a bit more detail. The Haber-Bosch process is a chemical process that is used to produce ammonia on a large scale. This process involves combining nitrogen and hydrogen gases under high pressure (100-200 atm) and temperature (450°C) in the presence of a catalyst, typically iron or ruthenium. As we can see from the chemical equation below (which you might recall from your high-school chemistry textbooks) three equivalents of hydrogen are needed for every nitrogen:
N₂ (g) + 3H₂ (g --> 2NH₃ (g)
It is the production of the hydrogen gas as the reactant that is responsible for 90% of emissions in ammonia production. The hydrogen is overwhelmingly (over 99%) produced from fossil fuels, with 70% from methane, via steam-methane reforming and 28% from coal gasification (using best available technology SMR has an emissions intensity of 1.8 tCO₂/tNH₃, compared to 3.2 tCO₂/tNH₃ for coal gasification). Since the Haber-Bosch reaction itself is exothermic (it releases heat), the energy input demands for heat, and subsequent emissions, are minimal in comparison. Emissions from the Haber-Bosch step (less than 50 MtCO₂e) come from the energy intensity of compressing the incoming nitrogen and hydrogen gases.
The production and application of ammonia-based fertilisers have a significant carbon impact in terms of the carbon emissions from nitrous oxide N₂O gases (GWP ~ 300 CO₂e) produced when fertilisers are applied to the soil. Ammonia is rarely (2%) the final product used to fertilise soils directly, it is instead converted to other fertiliser compounds including mostly urea and ammonium nitrate.
Ammonia production is highly centralised; there are currently only around 550 ammonia plants globally with ~250 Mt ammonia annual capacity in total (each plant produces between 600 and 3,300 tonnes per day). These very large scale Haber-Bosch plants are not currently designed to accommodate the variable pressures associated with renewables-powered electrolytic hydrogen. More localised on-demand ammonia production could facilitate decarbonisation, enabling colocation with renewable energy resources and helping to reduce over-fertilisation and subsequent N₂O emissions (notwithstanding the competing challenges of economies of scale and of building enhanced distribution). It is also a national security issue due to the interdependence of natural gas prices on fertiliser and therefore food costs.

An anhydrous ammonia plant. Source: WIRED
How big is the problem?
Ammonia production was 185 Mt in 2020, 70-80% of which is used as fertiliser. These fertiliser chemicals include urea (around 55% of fertilisers), nitric acid and ammonium nitrate (among others), with other applications in chemicals, plastics and textiles (acrylonitrile, melamine), in explosives, pharmaceuticals, and in refrigeration. Demand for ammonia for fertilisers alone is expected to grow from 156 Mt in 2020 to 267 Mt in 2050; this is in spite of the growing support for and awareness of increased nitrogen-use efficiency due to the emissions impact of fertiliser application.
There is expected to be growth in demand for ammonia as an energy carrier for use as a maritime fuel and in the power sector—it could represent a bigger proportion of demand than existing uses by 2050. Ammonia is proposed as an alternative fuel for ‘hard-to-electrify’ transportation sectors such as shipping (as well as heavy-duty trucking, aviation and even fuel for grid-scale energy production). It has two key advantages over proposed alternatives. Firstly, unlike alternatives such as methanol or e-fuels, it does not produce CO₂ emissions when burned or used in a fuel cell. And in comparison to hydrogen, it has much more favourable volumetric energy density, allowing it to be stored and transported more economically.
No ships to date are currently powered by ammonia, but it can in theory be used flexibly. It can be broken down, or ‘cracked’, into hydrogen and used in fuel cells or combusted. It is also possible to use ammonia directly, or to perform partial cracking and to use a mixture of hydrogen and ammonia. Indeed there is precedence for ammonia use as a fuel – during World War II buses based on an ammonia combustion engine were in use in Belgium as a response to shortages of liquefied fossil fuels.
Technologies and know-how exists already for its safe transportation and distribution — approximately 10% of ammonia is currently traded globally, so whilst this may not seem like a large proportion, it is nonetheless significant in terms of quantity (~20 Mt).
Decarbonising ammonia production
Today the hydrogen required for the Haber-Bosch process is typically produced via steam-methane reforming (SMR) which involves the reaction of methane with steam to produce syngas, which is a mixture of hydrogen and carbon monoxide, according to the equation:
CH₄ (g) + H₂O (g) --> 3H₂ (g) + CO (g)
This reaction is endothermic, and therefore requires an energy input. The syngas is then reacted with water further to product more hydrogen and CO2
CO (g) + H₂O (g) --> H₂ (g) + CO₂ (g)
Auto-thermal reforming (ATR) is a modification of SMR which also includes oxygen gas as a reactant. This produces carbon dioxide as an additional product, but the initial oxidation is exothermic and thus provides the heat required to the reaction for the breakdown of the methane. In essence, in ATR some of the methane is burned to provide the heat of the reaction, whereas in SMR the heat is provided externally. Since this produces a more concentrated stream of CO₂, it is considered to be more easily combined with CCS to produce 'blue' ammonia.
Decarbonising hydrogen production
The focus to date has been to move towards less carbon intensive hydrogen production. The main technologies currently available are to produce ‘green’ hydrogen via renewables-powered electrolysis or ‘blue’ hydrogen from fossil-fuel feedstocks with carbon-capture and storage (CCS). However currently neither of these technologies are deployed at scale and they are up to 2x more expensive to produce relative to conventional ammonia.
Although producing hydrogen for ammonia from methane with carbon-capture is likely to be the cheapest option in the short term, it will always be more expensive than conventional ammonia in the absence of carbon taxes/incentives. It also does not address the upstream emissions from fossil fuel extraction, including fugitive methane emissions.
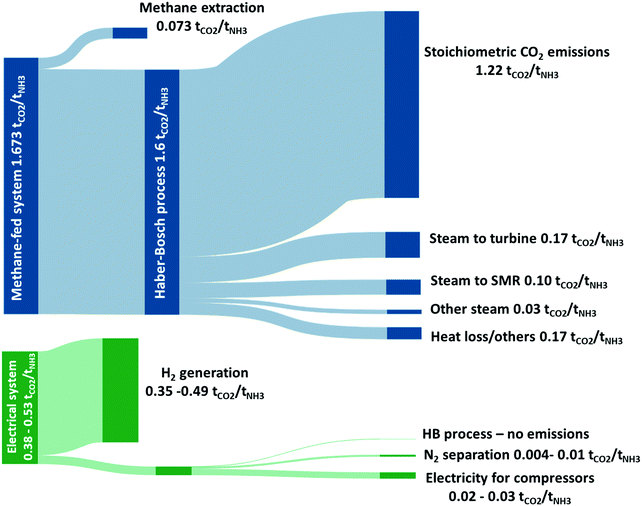
Sankey drawing comparing the attributions of direct CO₂-eq emissions arising from the methane-fed and the electrically driven Haber–Bosch processes (range of values depend on size of wind turbines). The stoichiometric CO₂ emissions are shown to highlight the minimum level of direct CO₂ emissions that can be achieved by the methane-fed system without carbon capture. The additional CO₂ emissions are allocated proportionally to the significant energy consumers. Source: C. Smith, A. K. Hill and L. Torrente-Murciano, Energy Environ. Sci. 13, 331 (2020)
Key challenges and opportunities
To produce green hydrogen at scale requires three key challenges to be overcome. Firstly we need lower cost hydrogen via electrolyser innovation to reduce costs and/or improve efficiencies – there is lots of exciting activity in this space. Secondly, the energy used to power the electrolysis must be truly low carbon – the energy intensity of hydrogen production is such that even a fairly 'low-carbon' grid could produce hydrogen that is dirtier than conventional grey hydrogen–the break even point is about 185 kgCO₂/MWh (i.e. on par with the UK grid in 2023). These risks have been well-documented elsewhere but must be kept front of mind. Finally, if dedicated renewable energy production is used for the hydrogen production then we must develop approaches to handle the intermittency: ideas include dedicated energy storage, buffering the hydrogen supply with a hydrogen tank or cannibalising some ammonia via cracking and returning it to the reactor to maintain a continuous process.
Direct electrochemical
Whilst largely still in the academic research phase, there is a growing body of research and startups tackling the direct electrochemical production of ammonia, where the energy for the chemical reactions is provided by electricity (rather than heat). Here, the nitrogen is taken from the air and the hydrogen is ultimately provided by water molecules. The reaction can either be direct, where the reaction occurs directly on the surface of the electrocatalyst, or indirect, where the reaction is mediated by another reaction.
Lithium-mediated electrochemical ammonia production
One of the most promising methods currently for electrocatalytic production of ammonia is the indirect method using lithium and ionic liquids. The lithium (Li) reacts with the nitrogen gas (N₂) to form the intermediate lithium nitride (Li₃N) on the surface of the Li electrode, which then reacts with a proton carrier to form NH3. This forms the basis for the technology currently being commercialised by Jupiter Ionics and Nitrofix Solutions. The key challenge with this approach is the high energy requirement; the necessary Li passivation step is a high voltage process, so the minimum thermodynamic voltage (and therefore input energy) is high.
Homogeneous electrochemical ammonia production
Molecular transition-metal complexes have emerged as effective catalysts for ammonia production, but here the electron transfer occurs in solution (homogeneous), rather than at the surface of the electrode (heterogeneous). Their molecular structure mimics the active site of the nitrogenase enzyme that naturally fixes nitrogen, which allows for good ammonia selectivity at moderate energy requirements, but within a smaller volume. The key technological barriers to overcome are the competing hydrogen evolution reaction (HER) which lowers the selectivity for ammonia formation (Faradaic efficiency), the mass transport of nitrogen within the system and catalyst stability.
Challenges and opportunities
The major challenge facing direct electrochemical ammonia synthesis is the strength of the N–N triple bond, which is energy-intensive to break (this is known as N2 activation). For electrochemical fuel production the technology targets to be achieved are those set by the Department for Energy's REFUEL programme (a current density of >0.3 A/cm2, 90% current efficiency (or Faradaic/Coulombic efficiency, 60% energy efficiency).*
The challenge is ensuring ammonia is produced selectively (high Faradaic efficiency) and in high yields instead of hydrogen via the hydrogen evolution reaction, and to do so with an energy efficiency that makes the process less (or comparably) energy intensive, and less costly, than producing green hydrogen followed by Haber-Bosch. Rigorous protocols of tests have been published for researchers to carry out to ensure that nitrogen reduction is indeed taking place. Results must be carefully verified and isotopically-labelled 15N2 gas used for quantification, largely due to the presence of ammonia or NOx contaminants in the air and/or lab system, which can produce false positives.
Example companies: Nitrofix (Israel), Jupiter Ionics (Australia), Nitrofix Solutions (Denmark), Atmonia (Iceland), SWAN-H (France).
Low-temperature thermochemical
There is increasing interest in designing a low temperature (<200°C) and low pressure (<10 atm) ammonia synthesis route. This would enable smaller, more distributed ammonia plants that are better adapted to intermittent generation of hydrogen from renewable-powered electrolysis. So why hasn’t one been designed to date?
The high-temperature, high-pressure HB process is restricted by the ‘scaling factor’. In order to reduce N₂ to make NH₃ the strong N–N triple bond must be broken. To do this, N₂ binds to the surface of the catalyst which breaks the bonds and allows N—H bond formation, NH3 then dissociates. Unfortunately, the strength of metal—N2 binding (which breaks the N—N bond, and is the rate determining step), is also related to the strength of metal—NH binding, and the two cannot be varied independently.
Significant progress has been made in recent years in understanding the role of surface structure and of different supports. New catalysts that are more performant at lower pressures are usually based on nanoclusters (only a few atoms across) of metal atoms, optimised with particular supports. This is the technology used by Starfire and Nium.
Challenges and opportunities
They key challenge here is to produce a suitable catalyst which good stability and reaction rate, that results in a levelised cost of ammonia (LCOA) that is competitive with that of conventional Haber-Bosch plants at scale.
Example companies: Tsubame BHB (Japan), Starfire Energy (USA), Liquium (New Zealand), ReMo (USA), Nium (UK).
Other early-TRL approaches include the use algae and bacteria for nitrogen fixation, but these techniques are unlikely to produce ammonia at scale (the footprint of the enzymes is too large), as well as photochemical ammonia synthesis (see Syzygy Plasmonics), chemical looping, plasma synthesis, nitride membrane synthesis and microwave ammonia synthesis (this is both more efficient, and also more tolerant to intermittent operation).
Enabling ammonia as a decarbonised energy carrier
Ammonia has been widely touted as a hydrogen carrier—in order to overcome the challenges of transporting hydrogen due to its low volumetric energy density it can be more readily transported as ammonia (which is liquid at moderate pressures, and has similar physical properties to propane, or LPG). The hydrogen can then be used as a fuel (for ships/heavy duty trucks/grid energy and so on). Since the ammonia can also be used directly as a fuel, it is perhaps more useful to think of it as an energy carrier. Either way, innovation is required to exploit ammonia's potential as an energy carrier and we discuss three areas of interest.
Ammonia cracking
Ammonia can be straightforwardly decomposed or ‘cracked’ into nitrogen and hydrogen gases, to then use the hydrogen as a fuel. It can be achieved above 700°C using inexpensive catalysts such as iron; lower temperature decomposition is possible involves the use of rare-metal catalysts such as ruthenium. There is active research (TRL 2-4) into the use of inexpensive sodium amides and imides that operate at ~500°C to achieve partial or complete cracking of ammonia.
The hydrogen produced can then be used in a fuel cell or hydrogen engine, as per the requirements for hydrogen (although PEM fuel cells are extremely susceptible to poisoning by any residual ammonia).
Example companies: Amogy (USA), Starfire (USA), OXGRIN (UK), Neology (Switzerland), Catalsys (UK), Sunborne (UK).
Ammonia fuel cells
Solid oxide and alkaline fuel cells are able to use ammonia directly as a fuel (without prior cracking to hydrogen). Solid oxide fuel cells can consist of oxide-conducting or proton-conducting solid electrolytes. Although solid oxide cells are more technologically mature, proton-conducting SOFCs have the added advantage of not producing NOx as a side product.
Example companies: ZEM fuels (UK), Faraday Fuels (USA).
Ammonia combustion engines
Ammonia can be used directly in an internal combustion engine (ICE) but has different combustion properties to petrol and hydrogen. To combat this typical ammonia-using engines usually require a combustion promoter, either in a dual-fuel system, or increasingly using hydrogen obtained via partial cracking of the ammonia to produce an ammonia/hydrogen mixture in-situ.
Example companies: Sunborne (UK), Catalsys (UK)

The ammonia-powered bus put into service in Belgium in 1943 (right) and its engine (which co-fired ammonia with fossil gas, left). Source: Kroch—Ammonia: a fuel for motor buses
Key challenges and opportunities
The opportunity for impact is big if we can successful transition away from current fossil-based shipping fuels. There are two main challenges, the first is a market challenge – ammonia is competing with methanol to replace bunker fuel and it is not yet clear whether one or the other, or both, will win the greatest market share. From a technical perspective the main challenges are minimising NOx emissions (including N2O), minimising ammonia slippage (unreacted ammonia) and developing combustion systems that can burn 100% ammonia.
Conclusion
Ammonia demand could more than double by 2050. And although we can minimise the impact of it's use as a fertiliser (N2O emissions); we will likely still need a significant amount of synthetic nitrogen fertilisers from ammonia. And so it is imperative that we decarbonise ammonia production as much as possible, first via green hydrogen production, then with the addition of modular electrochemical synthesis, or other innovative production technologies. Once we can scale up and bring down the cost of green ammonia, then we have the tantalising prospect of using it as a carbon-free fuel in many of our hard-to-abate sectors (shipping, aviation, heavy-duty trucking).
Footnote
Current density reflects the intrinsic rate of electron transfer between analyte and electrode surface, and depends of the electrode surface, the mass transport of the analyte towards the electrode and the kinetics of the electrode reaction.
Current efficiency (or Faradaic/Coulombic efficiency) is the ratio of actual product observed over the theoretical yield, essentially the reaction selectivity. A reaction with a low efficiency will have multiple side reactions and unwanted products.
Energy efficiency is the ratio of the theoretical minimum voltage required (thermodynamically) vs the actual applied voltage for the reaction. Additional electrical energy is often required to overcome activation energy—a good catalyst will improve the energy efficiency.
Further reading
Ammonia Technology Roadmap, International Energy Agency (2021)
Innovation Outlook: Renewable Ammonia, International Renewable Energy Agency and Ammonia Energy Association (2022)
For an excellent overview of ammonia utilisation technologies read: Green Ammonia Policy Briefing, Royal Society (2020)
Grand Challenges in Sustainable Ammonia Synthesis, DOE Roundtable Report (2016)
Ammonia Energy Association website, many
A zero emission blueprint for shipping, International Chamber of Shipping (2021)
The Future of Hydrogen, International Energy Agency (2019)
Challenges and prospects in the catalysis of electroreduction of nitrogen to ammonia, Bryan H. R. Suryanto et al. Nature Catalysis 2 , 290–296 (2019)
Excellent paper on technological milestones for green ammonia production: A Roadmap to the Ammonia Economy, Douglas R. MacFarlane et al., Joule 4, 1186–1205 (2020)
For an in-depth read about the chemistry of different approaches to next generation direct ammonia synthesis: Near ambient N2 fixation on solid electrodes versus enzymes and homogeneous catalysts Olivia Westhead et al., Nature Reviews Chemistry 7, 184–201 (2023)